exposure-attenuation quantities-and-units-used-to-describe-radiation-interaction cavity-theory
Atomic and nuclear structure - background
Plum-pudding model of the atom (Thomson model) (1904) = positive and negative charges uniformly distributed uniformly over the atomic volume
Rutherford model of the atom (1911) = mass and positive charge concentrated in the nucleus with electrons in a spherical cloud around it
Bohr model of the atom (1913) = electrons revolve around the nucleus in well-defined, circular orbits
- while in orbit, the electron does not lose energy despite being constantly accelerated
- angular momentum of the electron in the allowed orbit is quantized
- atom emits radiation only when an electron makes a transition from one orbit to another
notation
- X = atomic symbol
- A = atomic mass number (# of protons + # of neutrons)
- Z = atomic number (# of protons)
isotopes = Atoms which have the same number of protons but different number of neutrons isotones = Atoms which have the same number of neutrons but different numbers of protons isobars = Atoms which have the same number of nucleons (mass number) but different number of protons Isomers = Atoms with the same number of protons as well as neutrons. They are identical atoms that differ in their nuclear energy states
Classification of radiation
- Indirectly ionizing radiation = the energy is imparted to matter in a two-step process
- indirectly ionizing radiation transfers energy as kinetic energy to secondary charged particles, resulting in kerma
- charged particles transfer a major part of their kinetic energy to the medium, resulting in absorbed dose
- directly ionizing radiation = charged particles transfer a major part of their kinetic energy directly to the medium (resulting in absorbed dose)
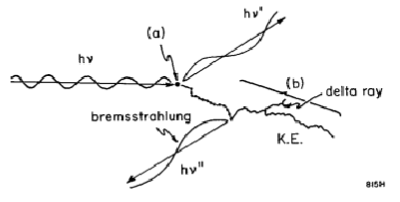
How is energy transferred from photons to the medium = the photon (hv) interacts with the medium at point (a) transferring energy to an electron as the photon is scattered (hv’); the transfer of energy is called kerma. The electron gives up most of its energy along path (b) in small collisions which is called absorbed dose. Along path (b), bremsstrahlung (hv”) results from the collisions of the electron with the nucleus. Delta rays may also result due to electron electron collisions.
4 categories of ionizing radiation
- characteristic x-rays = Results from electronic transitions between atomic shells.
- bremsstrahlung x-rays = Results mainly from electron-nucleus Coulomb interactions
- gamma rays = Results from nuclear transitions
- annihilation quantum = Results from positron-electron annihilation
| range | shielding | biological hazard | ||
|---|---|---|---|---|
| alpha particles | Large mass, highly charged, helium nuclei | 1-2 inches in air | dead layer of skin, paper | internal hazard. deposits large amount of energy in a small amount of tissue |
| beta particles | Small mass, electron size | short distance (1 inch to 20 feet) | plastic | internal hazard. externally, may be hazardous to skin and eyes. |
| neutrons | fairly large, no charge, has mass | very far in air (easily several hundred feet). High penetrating power is due to lack of charge | water, concrete, plastic (high hydrogen content) | external whole body exposure |
| gamma or x-rays | no mass, no charge, electromagnetic wave or photon | very far (easily several meters). high penetrating powe | water, lead | whole body exposure, hazard may be internal/external depending on where the source is |