Exposure attenuation = photons are attenuated via photon effect, Rayleigh, Compton scattering or pair production
attenuation = reduction in intensity or strength as it passes through the body
For a narrow beam fluence (I) of photons, incident on an absorber of thickness dx, the number of photons which interact with the target atoms in the absorber and which are removed from the beam is given by
Linear attenuation coefficient () = probability per unit path length that a photon will interact with the absorbing medium.
- It depends on the photon energy and the atomic number of the medium
Half value layer (HVL) = absorber thickness that attenuates the beam to 50% tenth value layer (TVL) = absorber thickness that attenuates the beam to 10%
Interaction of radiation with matter - Photon
Indirectly ionizing radiation occurs with photon beams
Summary:
| photoelectric effect | Compton effect | Pair production | Rayleigh scattering | |
|---|---|---|---|---|
| photon interaction | with whole atom (bound e-) | with free electrons (valance e-) | with nuclear coulomb field | with bound electron |
| mode of photon interaction | photon disappears | photon scattered | photon disappears | photon scattered |
| energy dependence | No | increases with energy | ||
| particles released | photoelectron | Compton electron | positron and electron pair | None |
| atomic coefficient dependence | ||||
| mass coefficient dependence | Independent | |||
| subsequent effect | characteristic x-rays, auger effect | characteristic x-rays, auger effect | annihilation radiation | None |
| Threshold | None | None | 1.02 MeV | None |
| significant energy region for water |
Photoelectric interaction
-
photon interacts with atom (tightly bound electrons) and ejects an orbital electron from the atom
-
process involves bound electrons (K, L, M, N shells)
-
entire energy of photon is transferred to the electron to eject it from the atom
-
kinetic energy of ejected photoelectron
-
emission of characteristic x-rays and auger electrons
-
atomic attenuation coefficient
-
mass attenuation coefficient
Compton interaction (incoherent scattering)
-
photon interacts with outer electron as though it were a free electron
-
photon transfers some energy to the electron ejecting it out of the atom with some KE
-
photon is scattered at an angle with a degraded energy
-
compton interaction is almost independent of atomic number
-
decreases with photon energy
-
amount of energy scattered and transferred depends on the angle of emission of the scattered photon and energy of photon
-
coefficient per electron / per gram
- for high Z materials
- for low Z materials
Pair production
- photon interacts with the electromagnetic field of an atomic nucleus
- photon gives up all of its energy in the process of creating a positron and electron
- threshold energy = 1.02 MeV
- photon energy in excess of the threshold is shared between the two particles as KE
- coefficient per atom (atomic attenuation coefficient)
- coefficient per gram (mass attenuation coefficient)
Triplet production (electronic pair production)
- photon disappears in the vicinity of an electron to produce two electrons and one positron
- threshold energy = 2.04 MeV
- Cross section for pair production and triplet production is zero for photon energies below the threshold energy, and increases rapidly for energy above the threshold
Rayleigh (coherent) scattering
-
interaction consists of an electromagnetic wave passing near the electron and setting it into oscillation
-
scattered x-ray has the same energy as the incident beam
-
probable in high atomic number materials with low photon energy
-
atomic Rayleigh cross-section
-
mass attenuation coefficient
Probability for an interaction
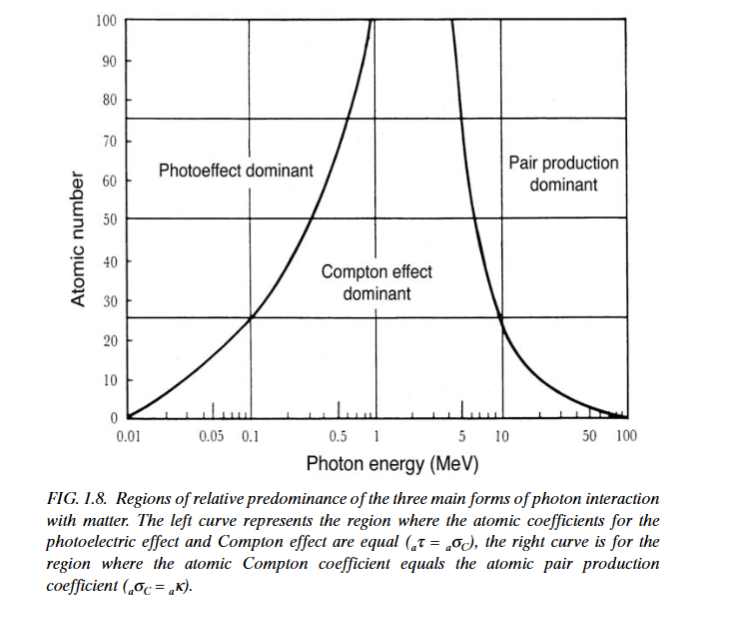
The probability of a photon to undergo any one of the various interactions depends on the energy of the photon and the atomic number
- photoelectric effect predominates low photon energy
- compton effect predominates intermediate photon energy
- pair production predominates high phootn energy
Interaction of radiation with matter - electrons
Inelastic collision with atomic electrons leads to
- atomic ionization = ejection of orbital electron from an absorber atom
- atomic excitation = transfer of an orbital electron from one allowed orbit (shell) to a higher level allowed orbit
Atomic ionization and excitation result in collision energy losses experienced by the incident electrons, and are characterized by collision (ionization) stopping power
Inelastic collision with nuclei result in bremsstrahlung (radiation loss) and is characterized by radiative stopping power